 
(Given the limits of hydrometry, I question whether the fifth digit matters much, and I am quite sure the sixth digit is noise.) My Handbook of Chemistry & Physics has at least three tables which give the density of water (one was for a gallon in pounds and grams, and I converted). SG values can be converted to metric density if you have actual value for water density at that time.The data is experimental, and different researchers have reported different results. SG values can be converted to metric density if you have actual value for water density at that time. not the typical distilled or deionised water used in chemical experiments!) Water density metrologists refer to Standard Mean Ocean Water (SMOW) which has with precise isotope levels and has undergone speciel purification processes (i.e. Even now, the density of water is vague as it depends on the water used for experiment. This is probably why some recommend a multipier of 0.999975 to conver SG 20/4 to metric density and others recommend 0.999973. I've since learnt that as the definition of the mL and 0 degree point on the Celcuis scale has changed between 19, so has the value for the density of water. I wanted the correlation between metric density and specific gravity 20/20 for sucrose in order to verify the Brix tables. I wasn't actually that interested in the Brix conversion as I had a table of Brix vs metric density (1998) and another table Brix vs specific gravity 20/4 and specific gravity 20/20 (1942). 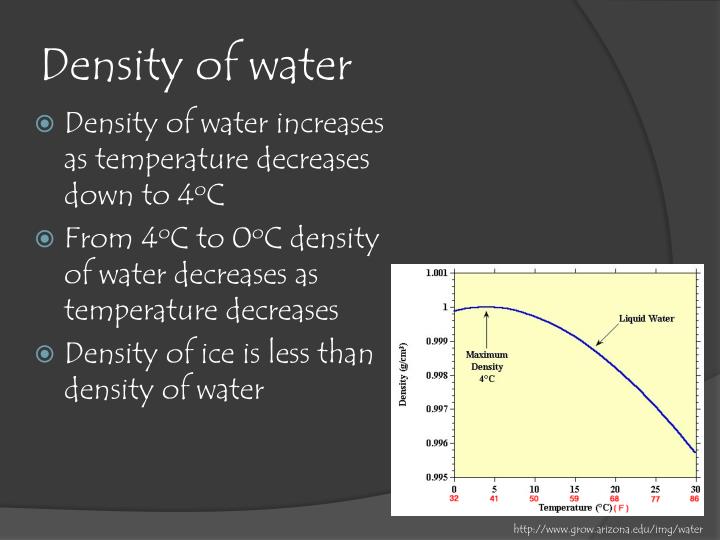
Thank you for the Brix thread and the useful information of the redefinition of the mL. Re: Metric density from specific gravity20/4, spec grav20/20 or apparent spec grav20/ 
There may be efffects, but I think they would be much smaller that 1 and 2 above. You can Google "air bouyancy" for techniques, formulas.ģ) Temperature: I don't know. The bouyancy effect of air on 1 L of sample is around 1 g, so this must be considered for density more precise than 0.001 kg/L. 
Particularly table 109 is in vacuo, but has typos, table 114 is apparent hence they are different. Be sure which you want agrees with which you are using. The NBS tables were published both for "in vacuo" and "apparent" data. Commercial weighing generally ignores it. High precision lab work considers this effect and reduces to "in vacuo" weight. On a balance, this affects both the weights and the sample. Use modern tables for density of water at 4 and 20 ☌Ģ) Just like water, air exerts a bouyancy effect on everything immersed in it (but much smaller). The SGs in the NBS tables should be unaffected, but the densities are obsolete. The modern definition of 1 dm³ gives a different absolute density. A few cautions.ġ) The liter had a difference definition in 1942 (1901-1960 really), 1 kg of water at 4 ☌. I put what little I could figure out in it. I'm grateful for any help I can get.We have a thread here on Brix. How are these factors calculated in the first place?ģ) Is the effect of redefinition of 0 'C in 1948 (IPTS-48), 1968 (IPTS-68) and 1990 (ITS-90) significant when converting older SG tables (probably based on experimental data with temp measurements using older temp scales) to metric density (SI)? This seems to suit the data well, but then I saw many other similar factors on the internet (none the same). Gupta (Practical density measurement and hydrometry,2002) multiplying SG 20/4 by 0.999975 produces metric density. Before I use tables/formula giving the metric density of sucrose solutions produced by an industry body (ICUMSA, 1998), I want to verify these values using brix tables published by NBS in 1942.ġ) What are the factors or equations to covert the following to metric density?Ģ) According to S.V. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
